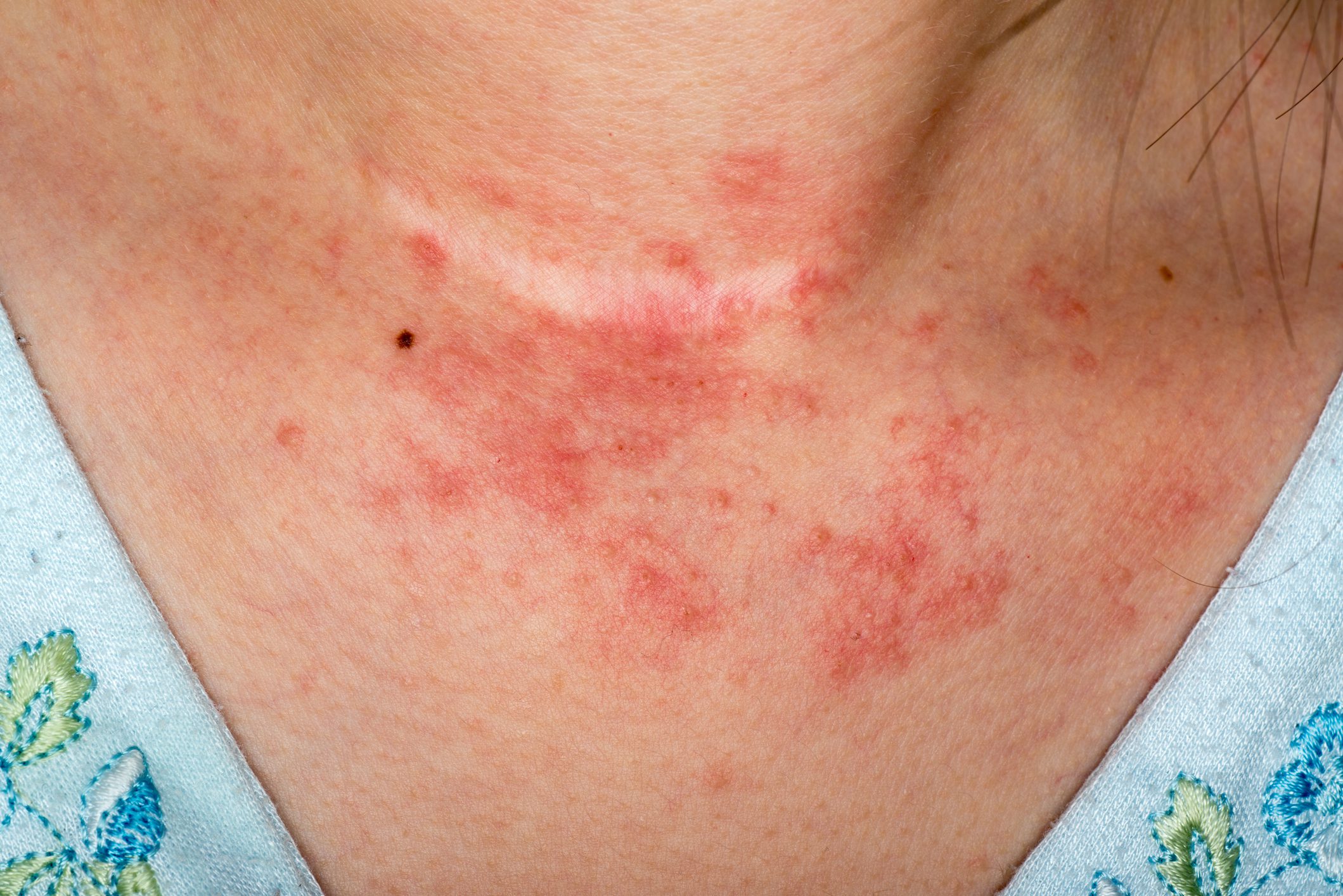
Although there have been no recent approvals of oral JAK inhibitors (JAK-i) for atopic dermatitis (AD), some interesting study findings have been published. These shed light on the safety of baricitinib, abrocitinib and upadacitinib with reference to the EMA opinion on JAK-i issued some time ago. In the EU, baricitinib has now been given an extended indication for AD patients aged 2 years and older.
Autoren
- Mirjam Peter, M.Sc.
Publikation
- DERMATOLOGIE PRAXIS
Related Topics
You May Also Like
- HER2+ early breast cancer
DESTINY-Breast11 – neoadjuvant T-DXd without anthracyclines
- Lavender aromatherapy
Take away the fear of the dentist
- Digital dermatology: innovative project examples
AI and eHealth tools in HS care
- Mantle cell lymphoma
Recurrence after BTK inhibition – prognostic factors and treatment options
- Dementia risk with type 2 diabetes
SGLT2 inhibitors with advantages vs. DPP4 inhibitors
- T helper 1 cells
Differentiation and activation of Th1 cells – a multi-omics approach
- Cannabis for schizophrenia
CBD pre-treatment could exacerbate side effects
- Type 2 diabetes: cardiovascular risk reduction is realistic