Local therapy with topically applied retinoids is a proven treatment strategy in acne patients. The latest member of this group of drugs is a trifarotene. The preparation is indicated for the external treatment of moderate acne vulgaris on the face and trunk and has shown promising results in clinical studies.
Acne vulgaris is an inflammatory disease of the sebaceous gland follicles of the skin that is particularly common in adolescents [1,2]. In addition to seborrhea, the central clinical sign is the appearance of efflorescences such as comedones, papules and pustules. The blockage of the sebaceous glands initially leads to the formation of microcomedones, from which skin-colored or whitish closed comedones form [3,4]. Further growth of the “blackheads” results in open comedones with a dark colored horny plug [4]. This stage is called acne comedonica [1]. Perifollicular inflammatory processes may result in papules and pustules [4]. This inflammatory form of acne is called acne papulopustulosa [1].
Topical retinoids: comedolytic and anti-inflammatory action.
Guidelines recommend topical retinoids for acne comedonica and mild acne papulopustulosa as a basic therapeutic [5]. Trifarotene (Aklief®) is a “first-in-class” representative of the fourth-generation topical retinoids and is indicated for the local therapy of acne vulgaris on the face and/or trunk in patients 12 years of age and older when many comedones, papules, and pustules are present [6]. Like other topical retinoids, trifarotene is a very potent comedolytic with anti-inflammatory effects. This topical retinoid is stable in keratinocytes for more than 24 hours and is rapidly metabolized by hepatic microsomes [7]. This allows it to exert an excellent effect in the skin, while the blood concentration is low [7].
Trifarotene: rapidly and persistently effective
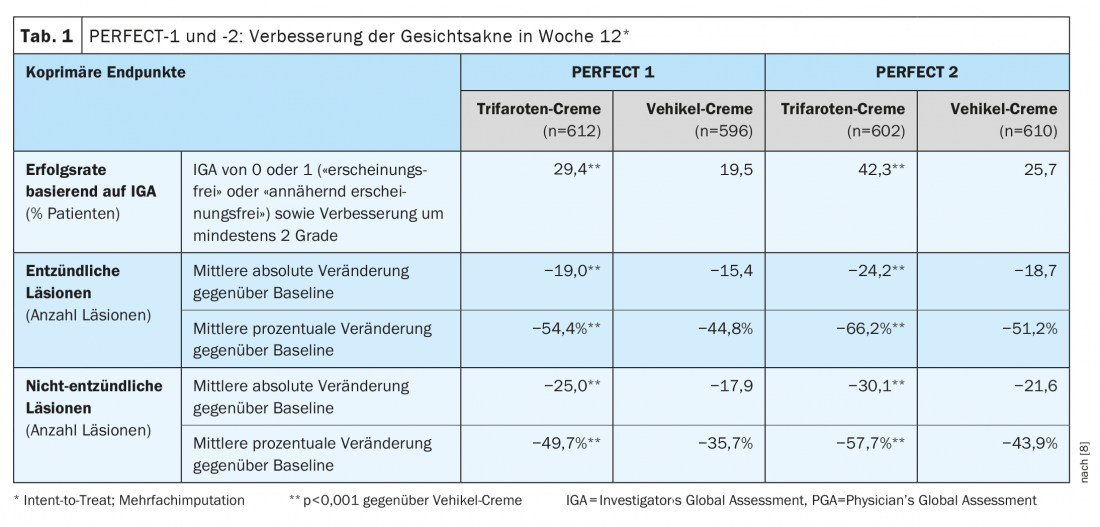
In both the PERFECT- 1 and 2 phase III trials, 1214 patients were treated with trifarotene, and 1206 patients received vehicle cream. At week 12, IGA success rates were 29.4% in PERFECT 1 and 42.3% in PERFECT 2 vs. 19.5% and 25.7% in the vehicle arm (p<0.001) [8]. Already at weeks 1 and 2, Trifarotene showed a significant reduction in the number of facial lesions compared with the vehicle group. The co-primary endpoints regarding improvement in facial acne at week 12 are shown in Table 1 [8]. Under trifarotene treatment, the mean absolute number of inflammatory lesions decreased by 19.0 and 24.2, respectively, compared with 15.4 and 18.7 in the vehicle group (p<0.001) and the mean absolute number of noninflammatory lesions decreased by 25.0 and 30.1 vs. 17.9 and 21.6, respectively, with vehicle (p<0,001). The observed side effects were consistent with the known safety profile of older generation retinoids. Irritation and itching at the application site and sunburn were most common [8,9].
The additional 52-week phase III study supported the results obtained in PERFECT-1 and -2 with a significant decrease in inflammatory and non-inflammatory acne lesions and an improvement in patient-reported quality of life [9].
Literature:
- Degitz K, Ochsendorf F: JDDG 2017; 15(7): 709-722.
- Bhate K, Williams HC: Br J Dermatol 2013; 168(3): 474-485.
- Gollnick HP, Zouboulis CC: Dtsch Arztebl Int 2014; 111(17): 301-312.
- Nast A, et al: Br J Dermatol 2016; 174(6): 1351-1358.
- Nast A, et al: S2k guideline on the therapy of acne. JDDG 2010; 8(Suppl 2): 1-59.
- Drug Information, www.swissmedicinfo.ch, (last accessed 05/25/2022).
- Aubert J, et al: Br J Dermatol 2018; 179: 442-456.
- Tan J, et al: JAAD 2019; 80(6): 1691-1699.
- Blume-Peytavi U, et al: JEADV 2020; 34: 166-173.
DERMATOLOGY PRACTICE 2022; 32(3): 28