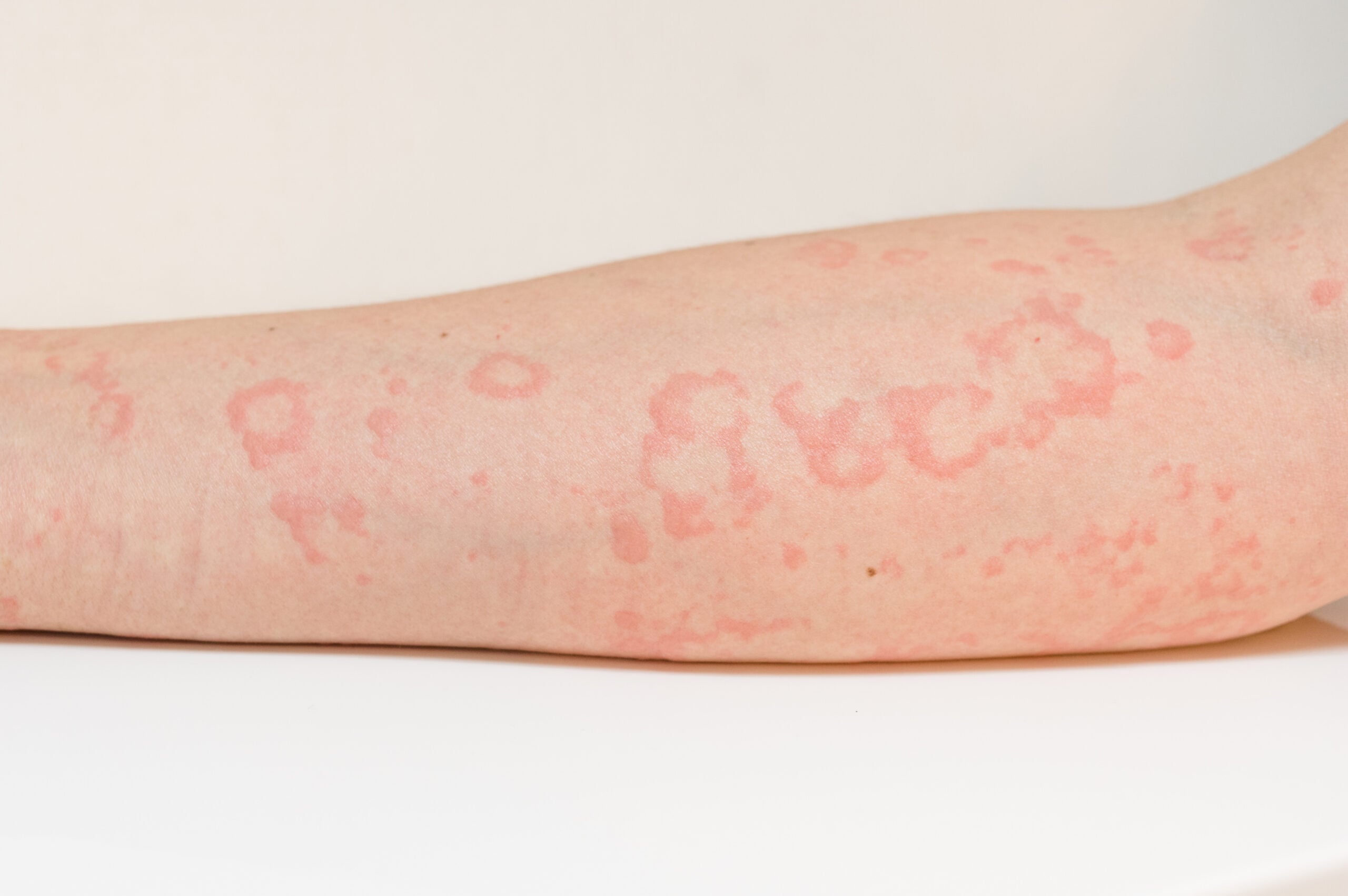
Chronic urticaria often leads to a high level of suffering, but new treatment options are on the way – a lot has happened since the publication of the current S3 guideline. Dupilumab is another biologic that has received FDA/EMA approval for CSU, but only for omalizumab-naïve patients. Remibrutinib, a small molecule, has also cleared the approval hurdles in the USA and new data is available on the biologics barzolvolimab and briquilimab. Total IgE is becoming increasingly important as a predictive biomarker for selecting the appropriate first-line systemic therapy.
Autoren
- Mirjam Peter, M.Sc.
Publikation
- DERMATOLOGIE PRAXIS
Related Topics
You May Also Like
- Proteins in wound healing
Do special amino acids lead to success?
- Psychedelic-assisted therapy
Current status of PAT
- Hepatitis B prophylaxis
Vaccination protection for healthcare staff with patient contact – an update
- Plastic surgery and reconstructive microsurgery for DFS
Functional limb preservation between infection control, vascular medicine and resurfacing
- Chronic urticaria: new treatment alternatives in sight
If antihistamines are not sufficient and IgE levels are low
- Adrenogenital syndrome
Clinical care from birth to adulthood
- Atopic dermatitis as a door opener for an atopic cascade?
AD trajectories: Summary of current findings
- New approvals, current study data and ADC development